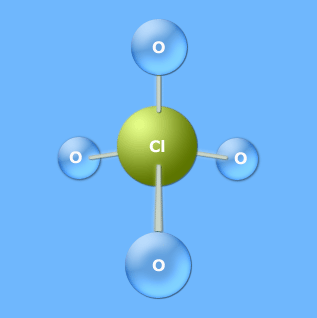
Sketch of ClO 3 - ion Lone pairs on atoms Therefore chlorine has moreĬhance to be the center atom (See the figure). To be the center atom, ability of having greater valance is important. For,ĬlO 3 - there are 26 valence electrons, so total pairs of electrons are 13. Total electron pairs are determined by dividing the number total valence electrons by two. Total valance electrons pairs = σ bonds + π bonds + lone pairs at valence shells Total valence electrons = 7 + 18 + 1 = 26.Due to -1 charge, received electrons to valence electrons= 1.Total valence electrons given by oxygen atoms = 6 *3 = 18ĭue to -1 charge, another electrons is added.There are three oxygen atoms in ClO 3 -, Therefore Total valence electrons given by chlorine atoms = 7*1 = 7.SoĬhlorine has seven electrons in its valence shell. Also there is a -1 charge onĬhlorine and oxygen are located at 7 and 6 groups respectively in the periodic table. There are one chlorine atom and three oxygen atoms in the chlorate ion. 
Total number of electrons of the valance shells of chlorine and oxygen atoms and charge of the anion Stability of lewis structure - Check the stability and minimize charges on atoms byĬonverting lone pairs to bonds to obtain the best structure.ĭrawing correct lewis structure is important to.Determine center atom from chlorine and oxygen atom.Total electrons pairs in valence shells.Oxygen atoms and including charge of the anion Find total number of electrons of the valance shells of chlorine and.Instead, it is often seen as a combination of various resonance structures.Following steps are required to draw ClO 3 - lewis structure and they are explained Furthermore, the chlorine atom is hypervalent. Furthermore, because all of the Cl-O bonds are the same length (1.49 in potassium chlorate), we cannot adequately describe it by one Lewis structure. The chemical formula of chlorate is ClO 3. Because of this, chlorates were once widely used in pyrotechnics, although their use has since declined due to their instability. Combinations of chlorates with almost any flammable substance (sawdust, sugar, organic solvents, charcoal, metals, etc.) can also explode. Because they are strong oxidizers, they should be stored away from organics and quickly oxidised materials. 
Sodium Chlorate is the most common type of chlorate (NaClO 3). They are extremely potent oxidising agents that are utilised in bleaching paper, herbicides, medicines, explosives, and other applications. In the lab, they may be made by adding chlorine to heated metal hydroxides such as KOH. Chlorate oxyanion is indicated by roman numerals in parenthesis, for example, Chlorate oxyanion (VII). Chlorine has an oxidation state of +5 in these ions.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
